Document Type : Original article
Subjects
Abstract
Background: Stem cell transplantation after acute myocardial infarction is a new therapeutic strategy that has been claimed to restore heart function. Some studies have reported that the cardioprotective effects of crocin are related to the regulation of the antioxidant enzymes activity and cardiac biomarkers. Accordingly, the aim of this study was to investigate the effect of the combined treatment with mesenchymal stem cells derived from adipose tissue and crocin in the isoproterenol-induced myocardial infarction model.
Methods: Forty rats were randomly divided into the control, acute myocardial infarction, acute myocardial infarction plus cellular therapy, acute myocardial infarction plus cellular therapy and crocin, and the group receiving crocin. Cardiac damage biomarkers and oxidative stress indexes such as, Malondialdehyde (MDA) and TACrolimus (TAC) levels were evaluated in all the groups.
Results: A significant elevation in serum concentrations of troponin T, creatine kinase, lactate dehydrogenase, and alanine aminotransferase were observed as the primary indicators of cardiac injury in the ischemia model group when contrasted with the control rats. These findings correlated with a marked reduction in total antioxidant capacity and a significant rise in MDA levels in the group treated with Isoproterenol compared to the control group. The administration of mesenchymal stem cells in conjunction with crocin demonstrated improvements in cardiac biomarkers, which were associated with a decrease in MDA levels and an increase in total antioxidant capacity.
Conclusion: Based on the results of this study, antioxidant compounds along with cell therapy is suggested as an effective therapeutic strategy in ischemic heart damage. Cellular and molecular studies are necessary to identify the exact mechanism of effectiveness of this therapeutic strategy.
Keywords: Crocin, Mesenchymal stem cells, Myocardial infarction, Rats
Introduction
Cardiovascular diseases encompass a broad spectrum of conditions, including cardiac ischemia, arrhythmias, and heart failure (1). Ischemic heart disease specifically refers to the damage of the heart muscle, known as the myocardium, resulting from inadequate blood and oxygen supply (2). This condition arises from a disparity between the oxygen supply and the heart muscle’s oxygen requirements. The primary underlying factor for ischemic heart disease is atherosclerotic disease affecting the epicardial vessels, where the accumulation of cholesterol and calcium within the arterial walls leads to their hardening and narrowing. This process ultimately reduces blood flow to specific regions of the heart muscle, resulting in ischemic damage to the myocardium (3).
Cardiovascular diseases represent one of the leading and most prevalent causes of mortality across all age groups and both genders. Statistics indicate that among the approximately 700 to 800 deaths occurring daily, over 300 can be attributed to these conditions, with heart attacks alone responsible for more than 150 of these fatalities (4,5). Despite significant progress in medical science and technology, the management of Myocardial Infarction (MI) continues to pose a complex challenge due to its multifaceted nature, which includes genetic factors, lifestyle choices, metabolic irregularities, and a complex interaction of molecular mechanisms. Traditionally, the primary objective in managing MI has been the swift restoration of blood flow to the affected coronary artery, thereby preserving the at-risk myocardium. The advent of Percutaneous Coronary Intervention (PCI) and thrombolytic therapy represented pivotal advancements in this area, resulting in enhanced patient outcomes and lower mortality rates. Nevertheless, these conventional methods are not without their drawbacks; for example, thrombolytic therapy carries a heightened risk of bleeding complications, and PCI may not always be an option due to delays in seeking care, logistical hurdles, or anatomical challenges. In recent years, there has been a notable shift in the approach to managing MI, propelled by a more profound understanding of its underlying mechanisms, innovative technological developments, and the integration of precision medicine strategies (7). The application of mesenchymal stem cells (MSCs) has demonstrated encouraging outcomes in MI by mitigating inflammation and enhancing cellular regeneration. Numerous studies have indicated that the utilization of these cells effectively contributes to the improvement of cardiac function, inhibits the enlargement of the infarct region, and promotes an increase in vascular density (8).
Numerous studies have reported that during MI, there is a marked reduction in antioxidant levels alongside an increase in oxidative markers, a phenomenon attributed to oxidative stress. Antioxidants are crucial in mitigating the harmful effects of free radicals during MI, and research has shown that a diet rich in antioxidants correlates with a decreased incidence of MI. However, mesenchymal stem cell transplantation can lead to adverse effects, primarily due to the unregulated suppression of immune cells. Furthermore, challenges such as the differentiation of transplanted cells into undesired cell types, the emergence of tumor-like cells, and the potential for metastasis to other tissues complicate this therapeutic approach. Additionally, a significant limitation of cell transplantation is the loss of over half of the transplanted cells either before or after they reach the site of injury (9,10). Consequently, the incorporation of adjuvant treatments that inhibit cell death may enhance the efficacy of cell therapy.
Research indicates that reactive oxygen species compromise the adhesion of MSCs, thereby diminishing the likelihood of recovery associated with this therapeutic approach. Consequently, it appears that the concurrent administration of antioxidant agents to neutralize free radicals may enhance the effectiveness of cell therapy. Thus, the objective of this study is to assess the impact of treatment involving adipose tissue-derived mesenchymal cells in conjunction with crocin in a rat model of isoproterenol-induced MI.
Materials and Methods
In this research, a total of forty male Sprague-Dawley rats, sourced from the Research Center at Ahvaz Jundishapur University of Medical Sciences in Iran, were utilized. These rats weighed between 170 and 190 gr and were aged between six to eight months. The rats were maintained in a controlled environment with temperatures ranging from 20-24°C, following a light cycle of 12/12 hrs of light and darkness, and were given unrestricted access to food and water for a duration of one week. Subsequently, the rats were randomly assigned to following groups.:
- Control: Healthy rats receiving normal saline.
- Acute MI: Receiving isoproterenol (subcutaneously, two consecutive days, 85 mg/kg and 170 mg/kg, respectively) (11).
- Acute heart infarction and cellular therapy: Receiving isoproterenol (subcutaneously, two consecutive days, 85 mg/kg and 170 mg/kg, respectively) with mesenchymal cells derived from adipose tissue, 24 hrs later of the second dose of isoproterenol.
- Acute MI and cellular therapy plus crocin: Receiving isoproterenol (subcutaneous, two consecutive days, 85 mg/kg and 170 mg/kg, respectively, with mesenchymal cells derived from adipose tissue systemically and 24 hrs after the second dose of isoproterenol + crocin (20 mg/kg intraperitoneally (12), for 2 weeks, starting 24 hrs after the second dose of isoproterenol.
Crocin: 20 mg/kg intraperitoneally (12), for 2 weeks.
Induction of MI
Isoproterenol was dissolved in normal saline and injected subcutaneously for two consecutive days with an interval of 24 hrs with a dose of 85 mg/kg and 170 mg/kg to induce acute MI based on previous studies. In order to confirm MI establishment, needle electrodes were carefully placed subcutaneously in the animals while they were under light ether anesthesia in the lead II configuration. Electrocardiogram (ECG) recordings were obtained via Bio-Amp monitored by a Power Lab system (AD-Instruments, Australia) and an increase in ST-segment considered as MI event according to the pervious documented studies (13,14).
Isolation of MSCs derived from adipose tissue
In this extraction technique, adipose tissue is isolated from the epididymis of a laboratory rat. The harvested adipose tissue is placed in a sterile Petri dish kept on ice, where it is meticulously cut into small fragments measuring approximately 1 to 2 mm3. These fragments undergo multiple washes with cold Phosphate-Buffered Saline (PBS) to eliminate blood cells and other residual tissue debris. Subsequently, the pieces are introduced into a falcon tube containing a 0.1% collagenase enzyme solution. Following the necessary duration for tissue digestion, DMEM culture medium supplemented with Fetal Bovine Serum (FBS) is added in a volume twice that of the enzyme to halt the enzymatic activity. After this step, the sample is centrifuged for 5 min at 2000 rpm. The supernatant is carefully removed and discarded, while the remaining liquid phase at the bottom of the falcon tube is transferred to a culture flask, to which a culture medium containing 10% FBS and 1% antibiotic (penicillin-streptomycin) is added. Once the cell density reaches 80% of the culture flask’s volume, Trypsin-EDTA is employed to detach the cells adhering to the flask’s bottom, which are then diluted at a ratio of 1:3 in DMEM culture medium with FBS. Depending on the quantity of detached cells, they are subsequently transferred to new culture flasks containing the same culture medium and returned to an incubator set at 37°C with 5% carbon dioxide. Initial observations are documented 24 hrs post-culture using a camera connected to an optical microscope (15).
Stem cells administration
In the current study, 24 hrs after acute MI model (injection of the second dose of isoproterenol), first the rats were anesthetized by a mixture of ketamine (50 mg/kg) and xylazine (5 mg/kg), then, the cells were injected with 106 MSCs systemically through the tail vein. On the 14th day after the injection of stem cells, the desired factors were evaluated (16).
Crocin treatment
Following establishment of the acute MI model, crocin was injected intraperitoneally for 2 weeks with concentration of 20 mg/kg (12).
Measurement of cardiac biomarkers
In this purpose, at the day 14th, after anesthetizing the animals, a blood sample was collected from the heart. Blood sample serum was separated by centrifuge and then applied to measure creatinine kinase, troponin T, lactate dehydrogenase, aspartate aminotransferase, alanine aminotransferase and alkaline phosphatase using special kits and via spectrophotometric method.
Measurement of Malondialdehyde (MDA) and Tacrolimus (TAC) levels
The cardiac tissue was separated and transferred to liquid nitrogen and kept at -80°C until the experiment. On the day of the experiment, the frozen tissues were carefully weighed and homogenized at a ratio of 1:10 in phosphate buffered saline, and then the samples were centrifuged at 12,000 rpm at 4°C for 15 min. The supernatant solution was applied to evaluate the oxidative stress indicators using assay kits via spectrophotometric method.
Data analysis
The SPSS software (V22.0) was used to analyze the obtained data. ANOVA statistical test and appropriate post hoc test were used to compare between groups if normal. Using the K_s test, the normality of the data was checked, and the equality of variances test was also reported. Data were calculated as Mean±SEM and p<0.05 was considered as a significant level.
Results
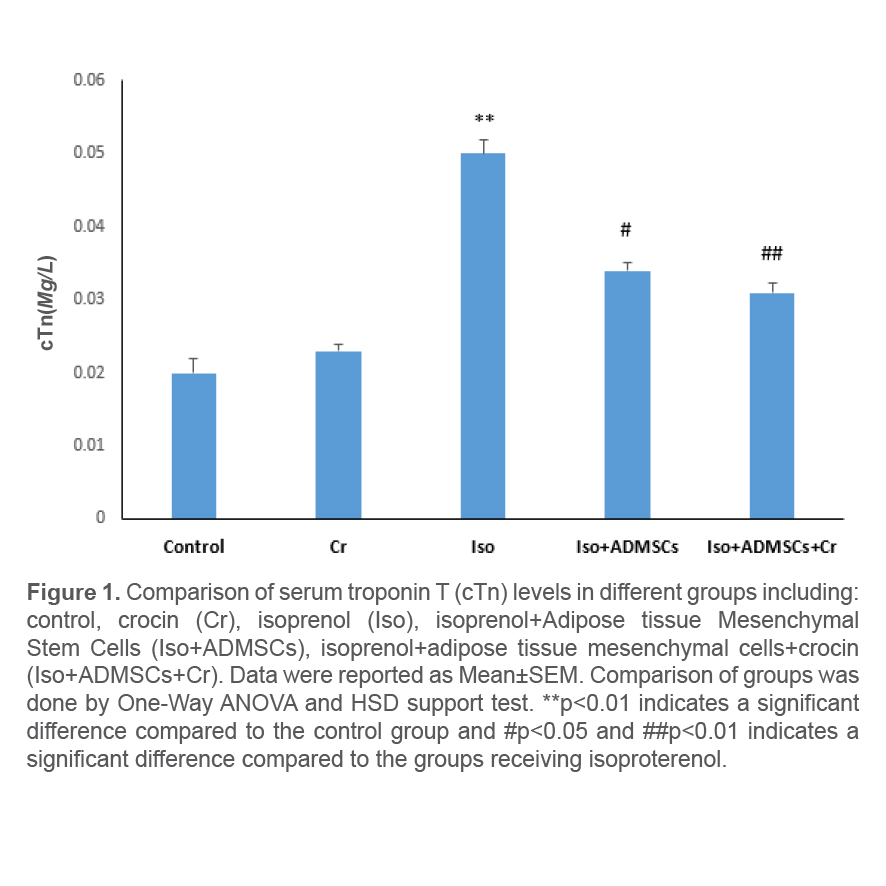
Effect of treatment with Adipose-Derived Stem Cells (ADSCs) and crocin on serum troponin T level
At the end of the study period, checking the serum level of troponin T in the studied groups showed a significant increase in the level of this enzyme in the groups receiving isoproterenol compared to the control group. In the group receiving ADSCs along with crocin, a significant decrease in serum troponin concentration was observed. This reduction was significantly higher than the group treated with ADSCs alone (Figure 1).
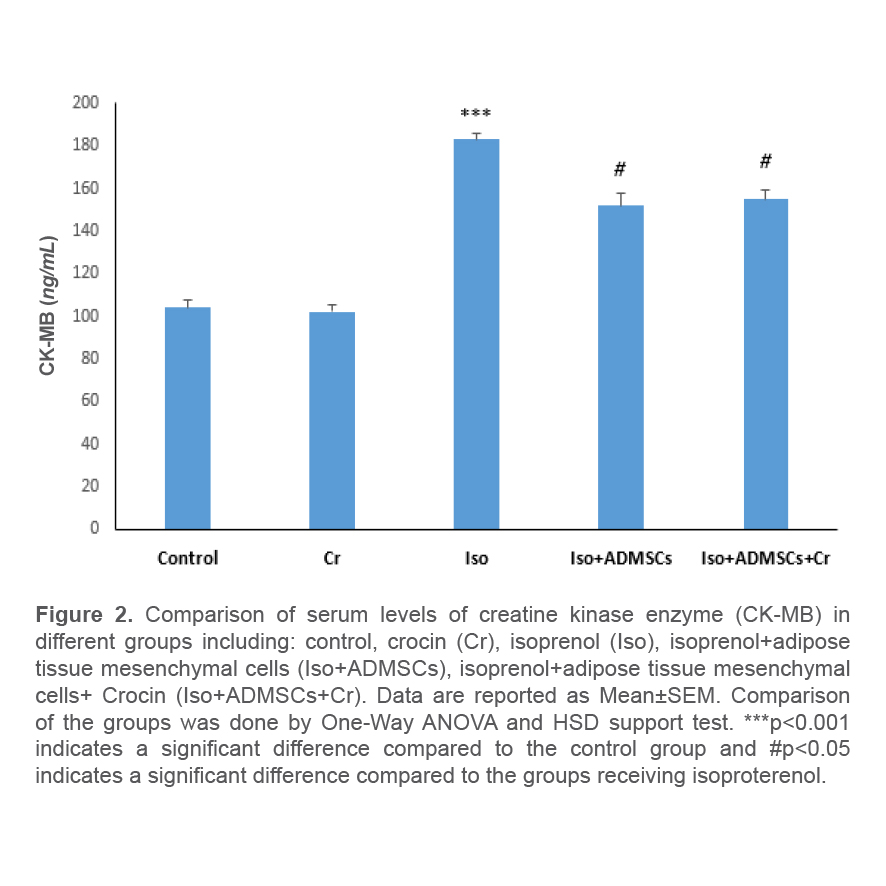
Effect of treatment with ADSCs and crocin on the serum level of creatine kinase enzyme
At the end of the study period, examination of the serum level of creatine kinase enzyme in the studied groups indicated a significant increase in the level of this enzyme in the groups receiving isoproterenol compared to the control group. In the group receiving ADSCs along with crocin, a significant decrease in serum creatinine kinase concentration was observed. This reduction showed no significant difference compared to the group treated with ADSCs alone (Figure 2).
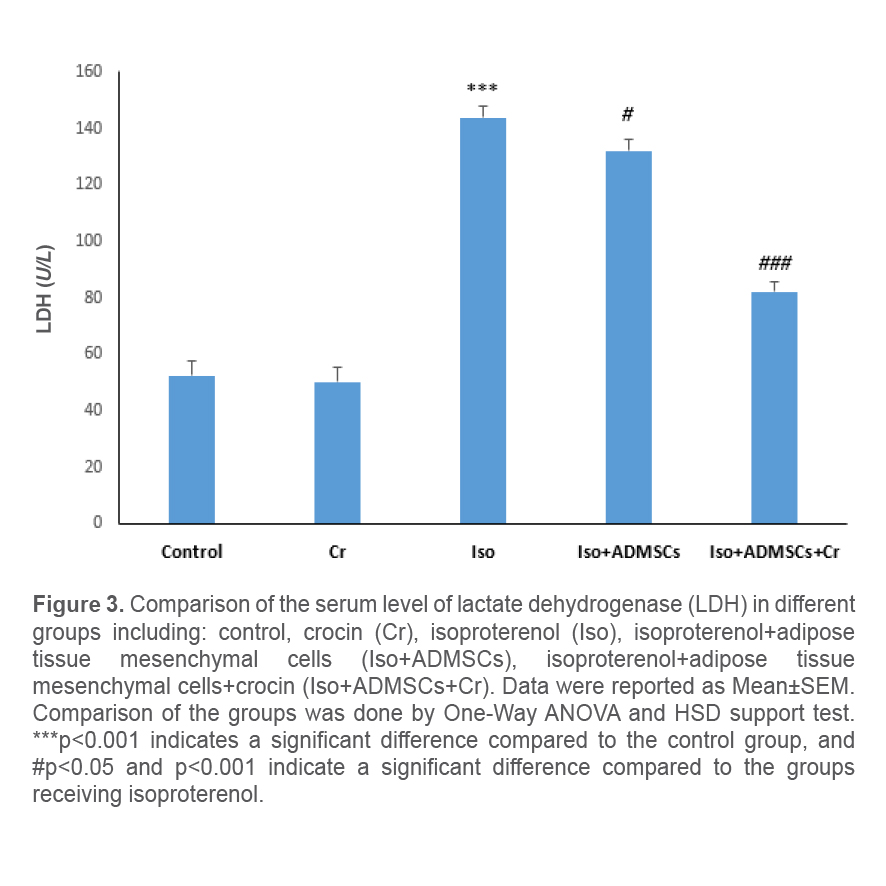
Effect of treatment with ADSCs and crocin on the serum level of lactate dehydrogenase enzyme
At the end of the study period, checking the serum level of lactate dehydrogenase enzyme in the studied groups demonstrated a significant increase in the level of this enzyme in the groups receiving isoproterenol compared to the control group. In the group receiving ADSCs along with crocin, a significant decrease in serum lactate dehydrogenase enzyme concentration was observed. This reduction showed a significant difference compared to the group treated with ADSCs alone (Figure 3).
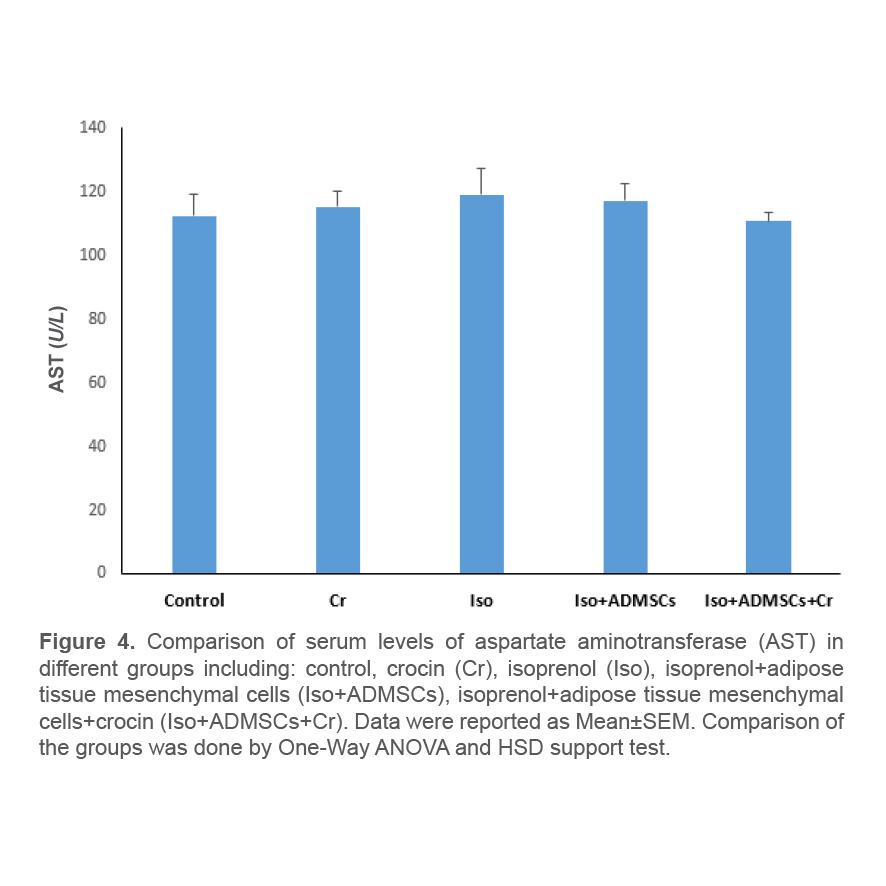
Effect of treatment with ADSCs and crocin on the serum level of aspartate aminotransferase enzyme
At the end of the study period, the examination of the serum level of aspartate aminotransferase enzyme in the studied groups represented no significant change in the level of this enzyme in the groups receiving isoproterenol compared to the control group. There was no significant difference in the concentration of this enzyme in the group receiving ADSCs together with crocin, as well as the group treated with ADSCs alone (Figure 4).
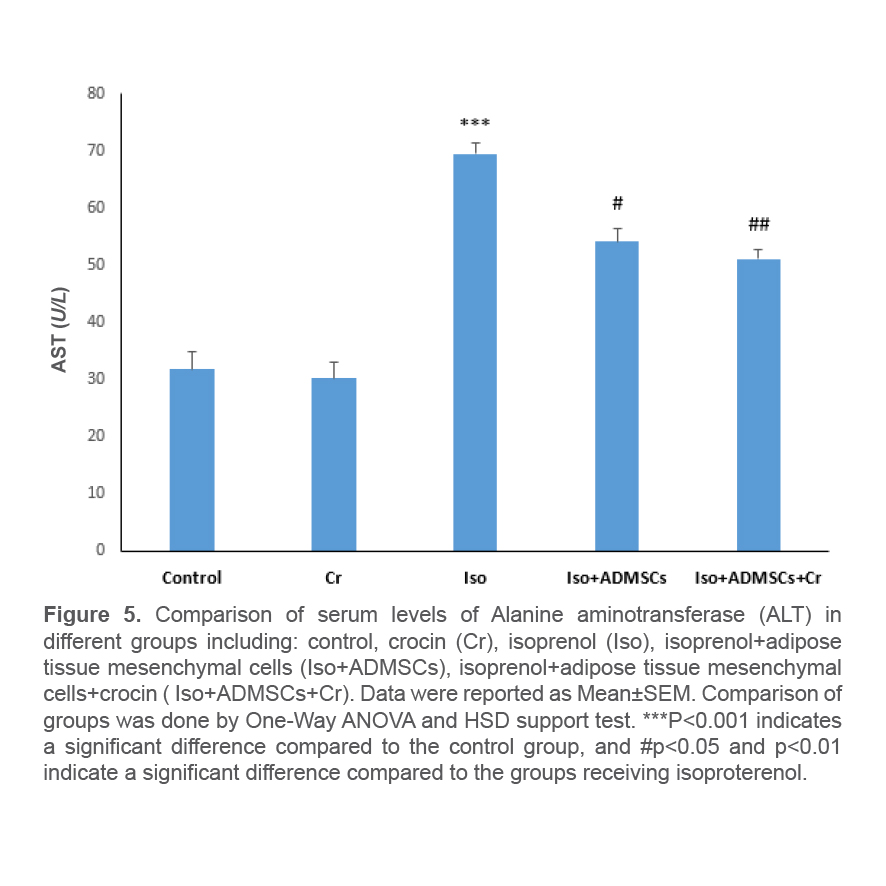
Effect of treatment with ADSCs and crocin on the serum level of alanine aminotransferase enzyme
At the end of the study period, checking the serum level of alanine aminotransferase enzyme in the studied groups showed a significant increase in the level of this enzyme in the groups receiving isopretronol compared to the control group. In the group receiving ADSCs along with crocin, a significant decrease in serum alanine aminotransferase enzyme concentration was observed. The reduction in the group receiving ADSCs together with crocin was significantly higher than the group treated with ADSCs alone (Figure 5).
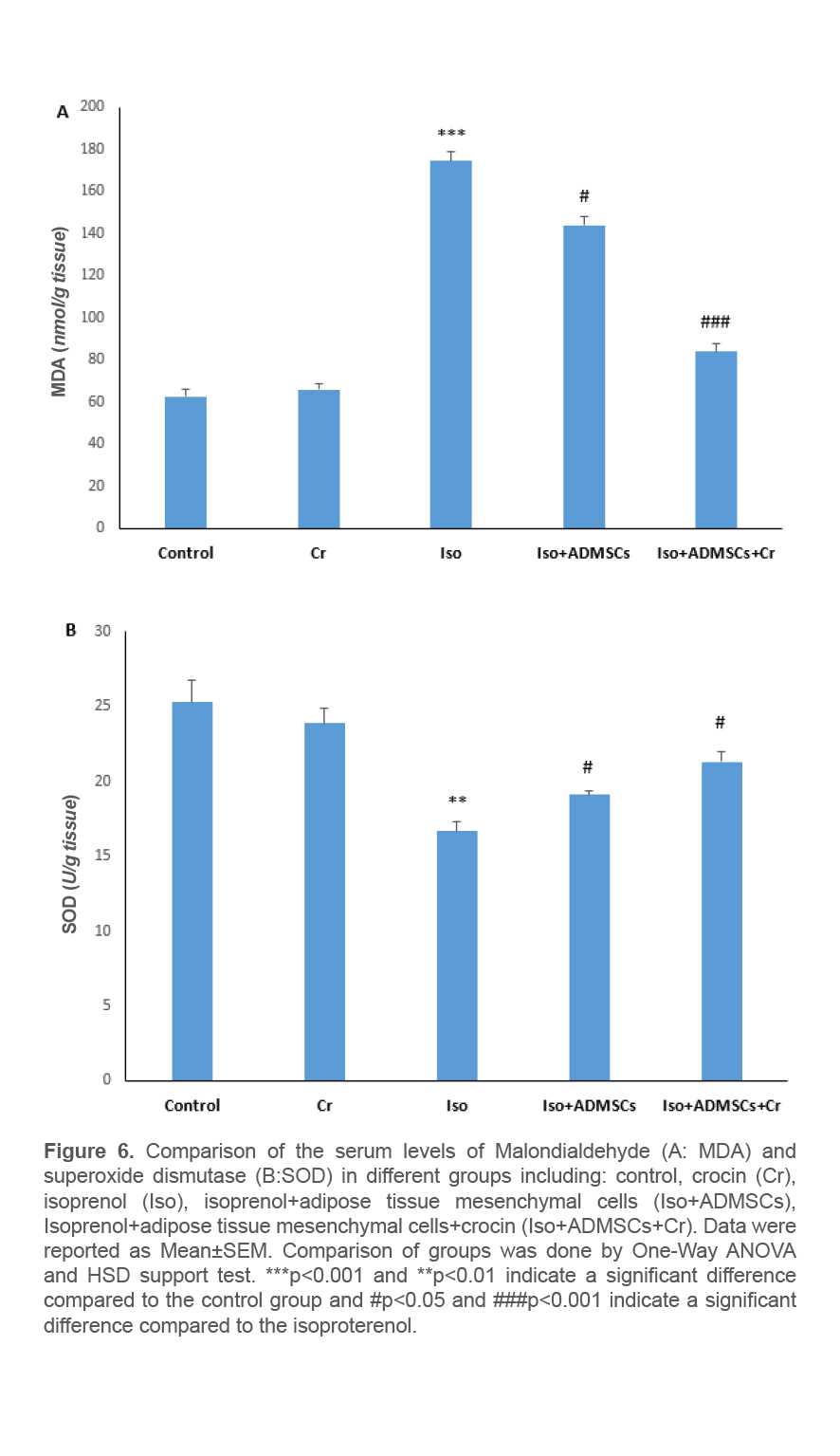
Effect of treatment with ADSCs and crocin on MDA and TAC levels in heart tissue
At the end of the study period, the examination of the MDA level in the heart tissue samples of the studied groups showed a significant increase in the level of this enzyme in the groups receiving isopretronol compared to the control group. In the group receiving ADSCs along with crocin, a significant decrease in MDA levels was seen. The reduction in the group receiving ADSCs with crocin was significantly higher than the group treated with ADSCs alone (Figure 6A). Examination of the TAC level in the heart tissue samples of the studied groups indicated a significant decrease in the level of this enzyme in the groups receiving isopretronol compared to the control group. In the group receiving ADSCs along with crocin, a significant increase in TAC was observed. The increase in the group receiving ADSCs together with crocin was significantly higher than the group treated with ADSCs alone (Figure 6B).
Discussion
The current research demonstrates the enhanced therapeutic efficacy of MSCs sourced from adipose tissue when combined with crocin, a recognized antioxidant, in addressing cardiac ischemia induced by isoprenaline administration in large laboratory rats. Ischemic Heart Disease (IHD) represents a critical cardiac disorder marked by diminished blood flow and subsequent cardiac hypoxia, serving as a significant factor in the broader context of Cardiovascular Disease (CVD) (17). This medical condition has the potential to result in heart failure and MI. Presently, ischemic heart diseases represent the leading cause of mortality in developed nations. Furthermore, they considerably contribute to the financial strain of healthcare systems in developing countries (18). Heart failure is particularly prevalent in industrialized regions, impacting approximately 3 to 5% of the global population, and has evolved into a progressive illness associated with substantial diagnostic expenses (19). Myocardial oxygen is essential for the survival and optimal functioning of the heart. Myocardial ischemia arises when the mitochondria fail to supply the necessary energy for the heart tissue (20). In such situations, the myocardium resorts to anaerobic glycolysis to generate energy. However, under anaerobic conditions, the myocardium’s capacity to produce sufficient oxygen to support cardiac cellular functions is compromised. This lack of oxygen results in diminished nutrient supply and insufficient removal of metabolic byproducts (21). This procedure modifies various components within the cardiac architecture, including endothelial cells, fibroblasts, vascular smooth muscle cells, and leukocytes. This phenomenon is a primary contributor to ischemic conditions and ischemic heart failure. The concept of cell-based therapy emerged in the scientific arena during the 1990s and was regarded as a potentially effective approach in regenerative medicine aimed at repairing cardiac injury following MI (22). MSCs have been suggested as a promising therapeutic approach for the regeneration of damaged cardiac structures and the repair of tissue through paracrine signaling (23). Furthermore, reports have indicated the efficacy of MSCs in treating various other cardiac conditions.
The current research demonstrated the notable therapeutic benefits of stem cells obtained from adipose tissue in a myocardial ischemia model using large laboratory rats. The therapeutic impact was evidenced by a marked reduction in the levels of troponin T, as well as serum concentrations of the creatinine kinase enzyme, lactate dehydrogenase, and alanine aminotransferase, which are critical indicators of cardiac injury. Furthermore, the enhancements observed in cardiac enzyme levels correlated with improvements in cardiac histological markers. The precise mechanism by which MSCs exert their therapeutic effects remains largely elusive. Numerous studies have explored the advantageous impacts of mesenchymal cell therapies, utilizing various agents both in vivo and ex vivo, particularly concerning cardiomyocytes. From these investigations, several potential mechanisms have been identified, including the engraftment and differentiation of mesenchymal cells into new cardiomyocytes, the role of signaling and paracrine mediators, the recruitment of endogenous cardiac stem cells to promote proliferation and tissue repair, as well as the stimulation of neovascularization and immune system modulation. Collectively, these four mechanisms work synergistically to significantly counteract the detrimental effects of ischemic injury on organ regeneration. The primary condition resulting from ischemic damage to the heart is the loss of contractile function in cardiomyocytes, which is further exacerbated by the replacement of healthy tissue with fibrotic scar tissue (24-26). Research in this domain indicates that the administration of stem cells produces an anti-fibrotic effect, which contributes to the reduction of scar size, facilitates the regeneration of injured tissue, diminishes inflammation, encourages cell growth and proliferation, and enhances blood vessel formation and perfusion, thereby aiding in the reversal of damage. Furthermore, findings from multiple studies reveal that MSCs possess notable anti-fibrotic, anti-inflammatory, and antioxidant characteristics (27,28). The findings of the present study align with those of previous research in this area; however, further molecular investigations are necessary to elucidate the precise healing mechanisms associated with stem cells.
The current investigation revealed that the administration of MSCs led to an enhancement in the oxidative stress markers within cardiac tissue. Oxidative stress is characterized by a disruption of the normal redox balance, resulting in an elevation of prooxidants or free radicals that can alter the structure of lipids, proteins, and DNA, ultimately inflicting damage on cells or tissues. Among the various free radicals studied, Reactive Oxygen Species (ROS) are the most prominent, encompassing reactive molecules that maintain a stable charge. The primary endogenous ROS include the superoxide anion (O2.-), the hydroxyl radical (•OH), and Hydrogen Peroxide (H2O2). 1. Superoxide anion (O2.-) is primarily generated by the enzymatic activity of the reduced Nicotinamide Adenine Dinucleotide Phosphate (NADPH)-oxidase (NOX) family or as a by-product of oxidative phosphorylation in mitochondria. The levels of mitochondrial-derived O2.- are influenced by various factors, including the availability of metabolic substrates, cytosolic calcium concentrations, pH levels, and oxygen tension (29). The O2.- produced through the electron transport chain complexes exhibits high reactivity, posing a risk of mitochondrial damage. The enzyme Superoxide Dismutase (SOD) facilitates the conversion of O2.- into hydrogen peroxide (H2O2). Nonetheless, H2O2 can also be synthesized through different metabolic pathways involving dual oxidases (DUOX). Although H2O2 is more stable than O2.-, its detoxification is crucial due to its weak peroxide bond, which makes it susceptible to reactions with metals like Fe2+, leading to the formation of reactive hydroxyl radicals via the Fenton reaction. Both H2O2 and O2.- are capable of diffusing across cellular membranes, potentially triggering cell death and inflammatory responses (30). Numerous investigations in this domain have demonstrated that MSCs possess the ability to diminish ROS and biomarkers indicative of oxidative stress (31). The therapeutic potential of MSCs has been explored across various disease models characterized by elevated ROS levels and signs of oxidative injury. For MSCs to deliver their therapeutic benefits, they must endure the challenging in vivo conditions following transplantation, which can complicate their administration (32). However, research indicates that MSCs exhibit a remarkable resilience to oxidative damage. Laboratory studies have revealed that MSCs can withstand oxidative and nitrosative challenges, a capability linked to heightened levels of antioxidant enzymes such as Superoxide Dismutase 1 (SOD1), Superoxide Dismutase 2 (SOD2), Catalase (CAT), and Glutathione Peroxidase (GPx). Furthermore, MSCs are known to express heat shock protein 70 (HSP70) and the sirtuin SIRT3, both of which may contribute to their resistance against oxidative and nitrosative harm. Additionally, SIRT1 is essential for the survival of stem cells in the presence of hydrogen peroxide (H2O2), and its increased expression has been shown to confer protective effects (33). SIRT6 has been suggested to promote resistance against oxidative damage and basal ROS production in MSCs by enhancing the expression of downstream antioxidant genes, such as heme oxygenase-1 (HO-1). The elevated levels of HO-1 contribute to the reduction of ROS production and the prevention of cellular senescence in MSCs lacking SIRT6, indicating its crucial role in the survival strategies of MSCs within oxidative conditions. Furthermore, MSCs not only utilize inherent antioxidants but also demonstrate considerable adaptability when faced with redox stress (34). MSCs that are subjected to Lipopolysaccharide (LPS), which induces the production of oxidative and nitrosative free radicals, have demonstrated a protective role (35). Furthermore, various adaptive mechanisms have been identified, including the upregulation and/or nuclear translocation of redox-sensitive factors such as Nuclear Factor kappa-B (NFκB), thioredoxin (TRX1), and apurinic/apyrimidinic endonuclease redox factor-1 (APE1/Ref). Research demonstrated that exposing MSCs to hypoxic conditions during cell culture can enhance their anti-inflammatory, antioxidant, and cytoprotective capabilities (36). The capacity of MSCs to withstand and react to oxidative stress may be essential for their effectiveness in transplantation and therapeutic applications at sites of tissue injury.
The current investigation represented that the combination of crocin and MSCs sourced from adipose tissue enhanced the efficacy of these cells in addressing ischemic injury induced by isoproterenol in the cardiac tissue of rats. This improvement was evidenced by a notable rise in superoxide dismutase levels and a reduction in malondialdehyde concentrations within the heart tissue. Crocin (C44H64O24) is one of the most valuable carotenoid compounds with various therapeutic properties. Numerous human and animal studies have reported beneficial preventive and therapeutic effects of crocin against health problems caused by oxidative damage (37).
In this context, a study conducted in vitro examining the impact of crocin on a cardiac ischemia-reperfusion arrhythmia model revealed that crocin effectively reduces the occurrence of ventricular arrhythmias. The findings indicated a notable decrease in both Ventricular Tachycardia (VT) and Ventricular Fibrillation (VF) among the subjects administered crocin. These observed anti-arrhythmic effects corresponded with an enhancement in the activity levels of the enzymes Catalase (CAT), SOD, and glutathione (GSH) in the crocin-treated group (38). The experimental investigation into heart hemodynamic parameters and infarct size within a rat model of ischemia/reperfusion revealed that crocin notably enhanced mechanical performance, as evidenced by the ±dp/dt metric, which serves as an indicator of left ventricular systolic and diastolic function. Additionally, there was an improvement in coronary flow and left ventricular systolic pressure. Notably, crocin treatment resulted in a reduction of the infarct area in the hearts of the rats studied (39). Furthermore, in ex vivo experiments involving isolated rat hearts that underwent 30 min of myocardial ischemia followed by 12 hrs of reperfusion, pretreatment with crocin led to a significant decrease in myocardial apoptosis and necrosis, while also enhancing left ventricular function (40).
Although earlier studies utilizing animal models have yielded encouraging outcomes, MSCs have yet to be deemed suitable for human clinical trials. Consequently, numerous strategies aimed at augmenting the therapeutic efficacy of MSCs continue to be explored. In this experiment, we propose a method to improve the therapeutic capabilities of MSCs in the context of cardiac regenerative therapy by leveraging the antioxidant properties of crocin. The findings indicate that crocin significantly enhances the therapeutic potential of MSCs for cardiac repair. Despite having attained some initial success, there are still several challenges to address, including issues related to scalability and clinical compatibility. Comprehensive future research is essential to create more innovative strategies that can effectively produce therapeutically viable human mesenchymal stem cells.
Acknowledgement
This study is part of Rozbeh Mirzae thesis (student of MD program). The authors acknowledge the grant obtained from Persian Gulf Physiology Research Center, Medical Basic Sciences Research Institute, the Ahvaz Jundishapur University of Medical Sciences, Ahvaz, Iran [Grant No. APRC-0101 IR.AJUMS.ABHC.REC.1401.008].
Conflict of Interest
There was no conflict of interest in this manuscript.