Document Type : Review article
Subjects
Abstract
Background: There is still disagreement about the prevalence and incidence of dysphagia in Parkinson’s Disease (PD). Since the prevalence of dysphagia has been reported very differently in the related literature, it is imperative to estimate pooled prevalence in PD patients. Thus, we conducted a systematic review and meta-analysis to estimate the pooled prevalence of dysphagia in PD overall and separately for each assessment method (subjective and objective).
Methods: These databases were searched: ScopusPubMed Web of Science ,and Google Scholar, January 1990 to October 2021. A random-effects model was used to pool the prevalence rates reported in the included studies by the Comprehensive Meta-Analysis (CMA( software .All designs of the studies were included.
Results: Thirty-nine articles entered the current meta-analysis. The global estimation of the overall prevalence of dysphagia in PD patients was 50.4% )95%CI:42.2-58.6) in all the 39 studies, which proved statistically heterogeneous (p<0.001). Moreover, the pooled prevalence by the subjective (30 studies) and objective (19 studies) methods of assessment were estimated to be 39.5 and 68.8%, respectively. There was also a significant relationship between the prevalence of dysphagia and age of Parkinson’s patients )r=0.44, p=0.011) and also between the prevalence of dysphagia and the duration of PD (r=0.55, p=0.006).
Conclusion: Based on the results, one in two patients with PD has dysphagia. This is proven that dysphagia is common in PD. The prevalence of objective dysphagia and subjective dysphasia were very different. It suggests that all Parkinson’s patients should be evaluated for swallowing disorders regularly with objective tools.
Keywords: Aphasia, Deglutition disorders, Humans, Incidence, Parkinson disease, Prevalence, Search engine
Introduction
Parkinson’s Disease (PD) is a common degenerative disorder in adulthood with the main motor and non-motor symptoms including rigidity, bradykinesia, resting tremor, postural instability, hypomimia, dysarthria, dysphagia, festination, freezing, dystonia, anosmia, and pain (1,2). The number of patients with Parkinson disease is projected to extend rapidly to about 12.9 million worldwide by 2040 (3). Swallowing is a physiological and biochemical process for human life leading to food and fluid being ingested safely. Dysphagia as a usual clinical swallowing problem in PD patients has an important role in their morbidity, mortality, and quality of life (4,5). In fact, the term dysphagia refers to the difficulty in the swallowing process (6). The swallowing process includes 4 phases: preoral, oral, pharyngeal, and esophageal (7). All four stages of swallowing can be impaired in PD (6-8). In general, dysphagia grows gradually in PD and there is a strong link between the incidence of dysphagia and disease progresses (9). Dysphagia can lead to negative consequences such as aspiration, pneumonia, and malnutrition in case it is not identified promptly in PD patients (7). Some symptoms of dysphagia usually appear in the early stages of PD (esophageal dysphagia) and some others usually appear in the advanced stages of PD (oral dysphagia and pharyngeal dysphagia) (6). Also, some symptoms of dysphagia may even go unnoticed until the final stages of PD and the patient may be unaware of them (6,7). Literature has shown the different prevalence of dysphagia ranging from 11.7 (10) to 100% (6) in PD patients. In the last systematic review in 2016, the rate of swallowing problems in Parkinson’s patients was reported to be between 11 and 80% (11). This systematic review and meta-analysis aimed to estimate the prevalence of dysphagia in Parkinson’s disease.
Materials and Methods
Search Strategy
All stages of implementation and writing of this systematic review were based on the PRISMA statement (12). We conducted a systematic literature search on October 15, 2021, in the PubMed, Scopus, Google Scholar, and Web of Science (From January 1, 1990, to October 15, 2021). The following keywords based on Medical Subject Headings (MeSH) were searched: prevalence and/or incidence, dysphagia and/or swallowing disorders and/or eating disorders, Parkinson’s disease or Parkinson, oropharyngeal dysphagia, esophageal dysphagia, swallowing and/or eating, deglutition or deglutition disorders. The search was performed without restrictions regarding types of dysphagia or types of assessment of dysphagia. Moreover, articles suggested by search engines were reviewed to identify further studies. Following duplicate deletion, screening the titles and/or abstracts was independently conducted by two authors to exclude obviously irrelevant papers. Following completion of the systematic searches discussed above, the authors also manually searched the reference list of eligible studies to further identify any papers not indexed in initially searched, resulting in one additional eligible study (13).
Eligibility criteria
All studies published between 1990 and October 2021 presenting the point of the prevalence of dysphagia in Parkinson’s disease were eligible for inclusion, with no geographic limitations, and no restrictions on disorders in a particular phase of swallowing. Only original studies (without limitations in study design) were included in this study, thus case studies, brief reports, systematic reviews, review articles, and posters were excluded. Data on humans of any age, gender, or race who had Parkinson’s-induced dysphagia at any location were searched, with no restrictions on the duration, severity, or age of onset of Parkinson’s disease. Individuals with histories of comorbid conditions affecting the swallowing (e.g., amyotrophic lateral sclerosis, multiple sclerosis, Dementia, and Alzheimer’s disease) were also excluded. Other inclusion criteria are as follows:
1) Using “Parkinson” in the title of the article, 2) Utilizing quantitative values in reporting the prevalence of dysphagia in the text, and 3) Only studies written in English were reviewed. Studies that were presented only as conference papers and abstracts were excluded.
Outcomes of interest
Outcomes investigated in this systematic review included the following:
1. Dysphagia or swallowing disorders as reported subjectively through interviews or questionnaires; 2. Dysphagia or swallowing disorders as detected objectively through clinical examination, Flexible Endoscopic Evaluation of Swallowing (FEES), Video Fluoroscopic Swallowing Study (VFSS), and Electromyography (EMG).
Data extraction process
All data were extracted from these studies by two authors (A.A and A.Sh) separately using a structural form. These variables were extracted: the name of the first author, geographical location, year of publication, the sample size, the mean age of the patients with PD, dysphagia diagnostic criteria (subjective or objective), the number of PD patients with dysphagia, and duration of PD. Also, these two authors separately assessed the quality of all studies before using them in the current study and agreed on data to be extracted. Any disagreements were solved by a third author.
Main and subgroup analyses
The main analysis involved the prevalence of dysphagia in Parkinson’s patients. Given that all stages of swallowing may be impaired in Parkinson’s disease (7,8), and that different symptoms of dysphagia (based on different phases of swallowing) usually occur at different stages of Parkinson’s disease (6,7), we decided not to consider any limitations in the swallowing phases, hence, to obtain the prevalence of dysphagia in this study, we considered disorders in any stage of swallowing as dysphagia. In the included studies, a variety of tools were used to evaluate and diagnose dysphagia (in different phases of swallowing) in Parkinson’s patients. To better standardize and interpret the results of these studies, we divided all the tools used in these studies into two groups: subjective methods including questionnaires, interviews, the Unified Parkinson’s Disease Rating Scale (UPDRS), Parkinson’s Disease Non-Motor Symptoms Questionnaire (NMSQuest), Scales for Outcomes in Parkinson’s disease (SCOPA-AUT), medical records, and EET-10 Scale. In this study, the patient’s self-report of dysphagia was considered as an interview (Table 1). The objective methods including VFSS, FEES, the swallowing speed, EMG, Manometry, Repetitive Oral Suction Swallow (ROSS) test, Modified Dysphagia Rating Scale (mDRS), and Chicago Assessment Scale (CAS). We conducted a meta-analysis to pool outcomes of the included studies, estimating the prevalence of dysphagia in Parkinson’s patients with 95% confidence intervals.
Table 1. Characteristics of the included studies
|
Study |
Location & date |
Study design |
Sample size (n) |
Mean age (year) |
Sex (male) |
Disease duration (M±SD) (year) |
With dysphagia (n) |
Tool of diagnosis |
Evaluation methods |
Prevalence (%) |
|
Edwards (23) |
USA 1992 |
Case–control |
98 |
67.7 |
59 |
NR |
51 |
UPDRS |
Subjective |
52 |
|
Bird (18) |
Austria 1994 |
Cross-sectional |
16 |
72.5±5.2 |
10 |
NR |
16 |
VFSS |
Objective |
100 |
|
Hartelius (16) |
Sweden 1994 |
Retrospective cohort |
249 |
NR |
129 |
NR |
102 |
Interview |
Subjective |
41 |
|
Nilsson (38) |
Sweden 1996 |
Cross-sectional |
75 |
71 |
43 |
9 |
65 |
ROSS test |
Objective |
87 |
|
Leopold (17) |
USA 1996 |
Retrospective cohort |
72 |
73±10 |
51 |
8.7±6.2 |
59 |
VFSS |
Objective |
82 |
|
Coates (35) |
UK 1997 |
Cross-sectional |
53 |
69.9 |
20 |
6.7 |
43 |
CAS |
Objective |
81 |
|
Fuh (24) |
Taiwan 1997 |
Cross-sectional |
19 |
68.4 |
15 |
3.17 |
12 |
VFSS |
Objective |
63.2 |
|
Fuh (24) |
Taiwan 1997 |
Cross-sectional |
19 |
68.4 |
15 |
3.17 |
6 |
UPDRS |
Subjective |
31.6 |
|
Clarke (34) |
UK 1998 |
Case–control |
64 |
66.7 |
39 |
9 |
46 |
Swallowing speed |
Objective |
72 |
|
Clarke (34) |
UK 1998 |
Case–control |
64 |
66.7 |
39 |
9 |
19 |
Interview |
Subjective |
30 |
|
Muller (28) |
Austria 2001 |
Retrospective cohort |
17 |
60 |
13 |
NR |
7 |
Medical records |
Subjective |
41 |
|
Volonté (33) |
Italy 2002 |
Cross-sectional |
65 |
66.3±9.1 |
29 |
6.6±4.1 |
23 |
UPDRS |
Subjective |
35 |
|
Volonté (33) |
Italy 2002 |
Cross-sectional |
65 |
66.3±9.1 |
29 |
6.6±4.1 |
46 |
mDRS |
Objective |
70 |
|
Siddiqui (45) |
USA 2002 |
Retrospective cohort |
44 |
66.5±9 |
24 |
8.3±6.5 |
13 |
questionnaire |
Subjective |
29.6 |
|
Potulska (6) |
Poland 2003 |
Case–control |
18 |
69.3±8.2 |
6 |
5.7±4.2 |
13 |
UPDRS |
Subjective |
72.2 |
|
Potulska (6) |
Poland 2003 |
Case–control |
18 |
69.3±8.2 |
6 |
5.7±4.2 |
18 |
EMG |
Objective |
100 |
|
Martinez (43) |
UK 2007 |
Cross-sectional |
525 |
67.6±10.4 |
304 |
7±5.3 |
147 |
PD NMSQuest |
Subjective |
28 |
|
Verbaan (44) |
Netherland 2007 |
Retrospective cohort |
420 |
61.1±11.5 |
269 |
10.5±6.5 |
231 |
SCOPA-AUT |
Subjective |
55 |
|
Cheon (21) |
Korea 2008 |
Cross-sectional |
74 |
64.9±8.6 |
28 |
6.4±6.1 |
24 |
PD NMSQuest |
Subjective |
32 |
|
Miller (27) |
UK 2009 |
Retrospective cohort |
137 |
73 |
85 |
5 |
115 |
Swallowing speed |
Objective |
84 |
|
Miller (27) |
UK 2009 |
Retrospective cohort |
137 |
73 |
85 |
5 |
51 |
UPDRS |
Subjective |
37 |
|
Barone (42) |
Italy 2009 |
Retrospective cohort |
1072 |
67.4±9.4 |
647 |
5.1 |
173 |
Interview |
Subjective |
16.1 |
|
Aydogdu (13) |
Turkey 2010 |
Case–control |
23 |
65.7 |
15 |
5.7 |
16 |
VFSS |
Objective |
69.5 |
|
Sung (47) |
Korea 2010 |
Case–control |
54 |
67.1±10.3 |
22 |
0.95±0.73 |
31 |
Manometry |
Objective |
67.4 |
|
Walker (41) |
UK 2011 |
Cross-sectional |
75 |
75±9.7 |
38 |
4.8 |
24 |
UPDRS |
Subjective |
32 |
|
Perez (48) |
France 2012 |
Retrospective cohort |
419 |
69±10 |
239 |
6±5 |
76 |
UPDRS |
Subjective |
18 |
|
Cersosimo (40) |
Argentina 2012 |
Case–control |
129 |
64.7±8.7 |
68 |
7.9±5.8 |
26 |
PD NMSQuest |
Subjective |
20.2 |
|
Auyeung (19) |
China 2012 |
Prospective cohort |
171 |
62.2±10.6 |
93 |
11.4±2.6 |
102 |
Interview |
Subjective |
60 |
|
Barichella (20) |
Italy 2013 |
Cross-sectional |
208 |
67.8±9.2 |
141 |
8.8±6.2 |
121 |
UPDRS |
Subjective |
58.2 |
|
Monteiro (37) |
Brazil 2014 |
Cross-sectional |
30 |
61.6±10.7 |
17 |
7.2±3.6 |
12 |
UPDRS |
Subjective |
40 |
|
Monteiro (37) |
Brazil 2014 |
Cross-sectional |
30 |
61.6±10.7 |
17 |
7.2±3.6 |
7 |
VFSS |
Objective |
22 |
|
Michou (26) |
UK 2014 |
Cross-sectional |
26 |
65±9 |
17 |
6.71 |
11 |
Interview |
Subjective |
42.3 |
|
Michou (26) |
UK 2014 |
Cross-sectional |
26 |
65±9 |
17 |
6.71 |
16 |
VFSS |
Objective |
61.5 |
|
Golabbakhsh (36) |
Iran 2014 |
Cross-sectional |
34 |
59.8±11.4 |
19 |
NR |
5 |
VFSS |
Objective |
14.7 |
|
Kim (25) |
Korea 2014 |
Case–control |
33 |
69.09±1.4 |
20 |
6±0.81 |
26 |
VFSS |
Objective |
78.8 |
|
Kim (25) |
Korea 2014 |
Case–control |
33 |
69.09±1.4 |
20 |
6±0.81 |
28 |
Interview |
Subjective |
84.8 |
|
Cereda (10) |
Italy 2014 |
Retrospective cohort |
6462 |
61 |
3772 |
7 |
754 |
PD NMSQuest |
Subjective |
11.7 |
|
Rajaei (39) |
Iran 2015 |
Retrospective cohort |
33 |
66.09 |
24 |
NR |
15 |
VFSS |
Objective |
45.45 |
|
Rajaei (39) |
Iran 2015 |
Retrospective cohort |
33 |
66.09 |
24 |
NR |
15 |
Interview |
Subjective |
45.45 |
|
Ou (29) |
China 2015 |
Cross–sectional |
518 |
61.9±10.6 |
- |
4.8±4.1 |
188 |
UPDRS |
Subjective |
36.3 |
|
Silverman (1) |
USA 2016 |
Cross–sectional |
68 |
68.7 |
55 |
7.8±8.2 |
36 |
Interview |
Subjective |
52.9 |
|
Nienstedt (46) |
Germany 2017 |
Case–control |
119 |
68.9±10.1 |
80 |
9.7±7.1 |
28 |
FEES |
Objective |
24 |
|
Su (31) |
USA 2017 |
Cross– sectional |
33 |
70 |
24 |
NR |
20 |
Interview |
Subjective |
62 |
|
Ding (22) |
China 2017 |
Retrospective cohort |
116 |
64.1±9.2 |
70 |
NR |
101 |
VFSS |
Objective |
87.1 |
|
Ding (22) |
China 2017 |
Retrospective cohort |
116 |
64.1±9.2 |
70 |
NR |
36 |
Interview |
Subjective |
31 |
|
Pflug (4) |
Germany 2018 |
Case–control |
119 |
68.9±10.1 |
80 |
9.7±7.1 |
113 |
FEES |
Objective |
95 |
|
Umay (9) |
Turkey 2019 |
Case–control |
120 |
63.3±8.6 |
42 |
9.2±5.1 |
63 |
Eat-10 score |
Subjective |
52.5 |
|
Polychronis (30) |
UK 2019 |
Retrospective cohort |
398 |
61.6 |
260 |
0.54±0.53 |
49 |
SCOPA-AUT |
Subjective |
12.3 |
|
Wamelen (32) |
UK 2020 |
Retrospective cohort |
291 |
68.9±10.7 |
185 |
8.9±5.3 |
127 |
PD NMSQuest |
Subjective |
43.6 |
|
Total |
1990–2020 |
- |
13038 |
66.7±0.7 |
374 |
6.84±0.7 |
3326 |
- |
- |
50.4 |
NR= Not Reported, UPDRS=the Unified Parkinson’s Disease Rating Scale, ROSS-test=Repetitive Oral Suction Swallow, CAS=Chicago Assessment Scale, PD NMSQuest=Parkinson’s Disease Nonmotor Symptoms Questionnaire, VFSS=Video Fluoroscopic Swallowing Study, EMG=Electromyography, FEES= Flexible Endoscopic Evaluation of Swallowing, SCOPA-AUT=Scales for Outcomes in Parkinson’s disease, mDRS=Modified Dysphagia Rating Scale.
Statistical Analysis
The prevalence of dysphagia for each study was calculated based on the number of this event in relation to the sample size. Therefore, we calculated the estimates of the pooled prevalence of dysphagia weighted by sample size. Prevalence was reported with forest plots produced for all prevalence estimates, with 95% Confidence Intervals (CI). Due to the heterogeneity of the outcomes of included studies, a random-effects model was used to combine the outcomes of these studies. Thus, after calculating the prevalence of dysphagia for each study, by the comprehensive meta-analysis software (version 2.0, USA) (14), we performed a random-effects meta-analysis. Random-effects meta-analyses account for the heterogeneity of included data in case they are really related (15). When studies were statistically heterogeneous, the random-effects model was used to account for between-studies variation. We also consider a heterogeneity statistic (Q) for each of these analyses, which was utilized to calculate I2. The I2 allows us to examine whether the percentage of observed variance is real or spurious. The low percentage of I2 indicates that the most variance is spurious (15).
Results
Study selection
Systematic searches yielded 6,419 publications, as shown in the PRISMA Figure (Figure 1). In duplicate deletion, 5671 repetitive records were excluded. 749 studies entered the systematic stage. After analyzing the title and abstract of these studies, 565 irrelevant studies were excluded. The authors examined the full-texts of 184 articles, 141 articles that did not fulfill the inclusion criteria were excluded and 4 articles were excluded due to insufficient data. Also, the authors identified one eligible article from the reference list of review articles (13). Finally, 39 eligible studies entered the meta-analysis stage (Figure 1).
Characteristics of the included studies
Table 1 summarizes the characteristics of the included studies. As shown in table 1, we extracted 49 records from 39 studies. Over a third of the included records (n=17) were cross-sectional studies (43.6%), 35.9% (n=14) were retrospective cohort studies, 30.7% (n=12) were case–control studies, and one study was prospective cohort. The included studies in this systematic review were conducted in 16 different countries, the frequency of study locations included Europe (n=8; 50%), Asia (n=5; 31%), and America (n=3; 19%). In detail, of the 39 included studies, eight were conducted in the UK, five in the USA, four in Italy, three in Korea, three in China, two in Iran, two in Sweden, two in Austria, two in Germany, two in Turkey, one in Taiwan, one in Argentina, one in the Netherlands, one in France, one in Poland, and one was conducted in Brazil. These included studies were reported between January 1991 and March 2021.
A total of 13038 PD patients were extracted across 49 records. Of this population, a total of 58.9% of these Parkinson’s patients were male (n=7374). The pooled mean age of PD patients was 66.7±0.75 (range: 59.8–75) years, although one study did not report details of age (16). The pooled mean of the duration of Parkinson’s disease was 6.84±0.73 (range: 0.54–11.4) years, although 20% (n=8) of the studies did not report details of the duration of PD (Table 1).
Most of the included studies considered and reported oral, pharyngeal, and esophageal dysphagia as dysphagia. One study also claimed that the questionnaire it used identified signs of abnormalities in all stages of swallowing, but did not describe the name and characteristics of this questionnaire (17). A majority of records (n=30; 61%) employed the subjective evaluation methods (such as questionnaires, Interviews, and medical records) as assessment tools, and 39% of the studies (n=19) employed the objective evaluation methods (such as FEES, VFSS, EMG, and Manometry). In detail, out of 30 records that used subjective evaluation methods, one-third of the studies (n=10, 33%) used the UPDRS, one third of the studies (n=10, 33%) utilized an interview, 5 of them used the PD NMSQuest, two used the SCOPA-AUT, one used the EAT-10, one used the medical records and one used another questionnaire. Also, out of 19 records that used objective evaluation methods, more than half of the studies (n=10, 52%) used the VFSS, two studies used the FEES, two studies used the Swallowing speed, one of them used the EMG, one used the manometry, one utilized the ROSS test, one used the mDRS and one used the CAS. 10 studies reported both subjective and objective outcomes (Table 1). The highest prevalence of dysphagia in PD was reported in Austria (18) and Poland (6) (100%) and the lowest prevalence was reported in Italy (11.7%) (10).
Based on the results of most included studies (32 records), there was a significant relationship between the prevalence of dysphagia and the age of PD patients (r=0.44; p=0.011), as well as the prevalence of dysphagia and the duration of PD (r=0.55; p=0.006). Also, there was a significant correlation between subjective dysphagia and duration of PD according to 18 studies (r=0.65; p=0.015) (1,9,16,19-33), and objective dysphagia correlated with the age of PD patients according to 14 studies (r=0.82; p=0.000) (4,13,17,18,22,25,26,33-39).
Prevalence in the reviewed outcomes
To estimate the pooled prevalence of dysphagia in patients with PD, the results of the meta-analysis were obtained by the random-effects model. Based on estimates from 39 studies (n=13038 patients) (1,4,6,9,10,13,16-48), the overall prevalence of dys-phagia in PD patients was 50.4% (95% CI: 42.2-58.6) (Figure 2).
The highest prevalence of dysphagia in PD was 100% related to the study of Bird et al (18), and the lowest prevalence of dysphagia in PD was 11.7% associated with the study of Cereda et al (10). According to the dysphagia evaluation methods, the prevalence of dysphagia in PD by the subjective methods from thirty studies (n=11932 patients) (1,6,9,10,16,19-21,23,24,27-32,34,37,40-46,48) was 39.5% (95% CI: 31.2-48.4) (Figure 3).
In subjective evaluation methods, the highest prevalence of dysphagia in PD was 84.8% related to the study of Kim et al (25), and the lowest prevalence of dysphagia in PD was 11.7% found in the study of Cereda et al (10). The prevalence of dysphagia in PD by the objective evaluation methods from 19 studies (n=1106 patients) (4,6,13,17,18,22,24,25,27,29,33-39,47) was 68.8% (95%CI: 54.9-80) (Figure 4).
In objective evaluation, the highest prevalence of dysphagia in PD was 100% related to the study of Bird et al (18), and the lowest prevalence of dysphagia in PD was 14.7% found in the study of Golabbakhsh et al (36).
Trend of prevalence
In this study, we also looked at the progress of Parkinson’s dysphagia over the past 30 years. Most studies were from the last 10 years (2011 to 2021) with 26 studies (Table 1). The prevalence in the last 10 years (2011 to 2021 was 44.7%) is less than the last 10 years of the 20th century (1991 to 2000 was 64/4%). Table 2 contains a summary of the prevalence of dysphagia in PD over three decades from 1990 to 2021.
Table 2. Three types of dysphagia prevalence in PD from 1990 to 2021 and in total (N=13038)
|
Rate of prevalence * |
Period of time |
||
|
Overall dysphagia (%) |
Objective dysphagia (%) |
Subjective dysphagia (%) |
|
|
64.4 |
79.4 |
40.1 |
1990-2000 |
|
50.1 |
69.3 |
40.7 |
2001-2010 |
|
44.7 |
57.3 |
39.1 |
2011-2021 |
|
50.4 |
68.8 |
39.5 |
Total (1990-2021) |
*All prevalence rates are estimated through meta-analysis (Comprehensive Meta-Analysis software).
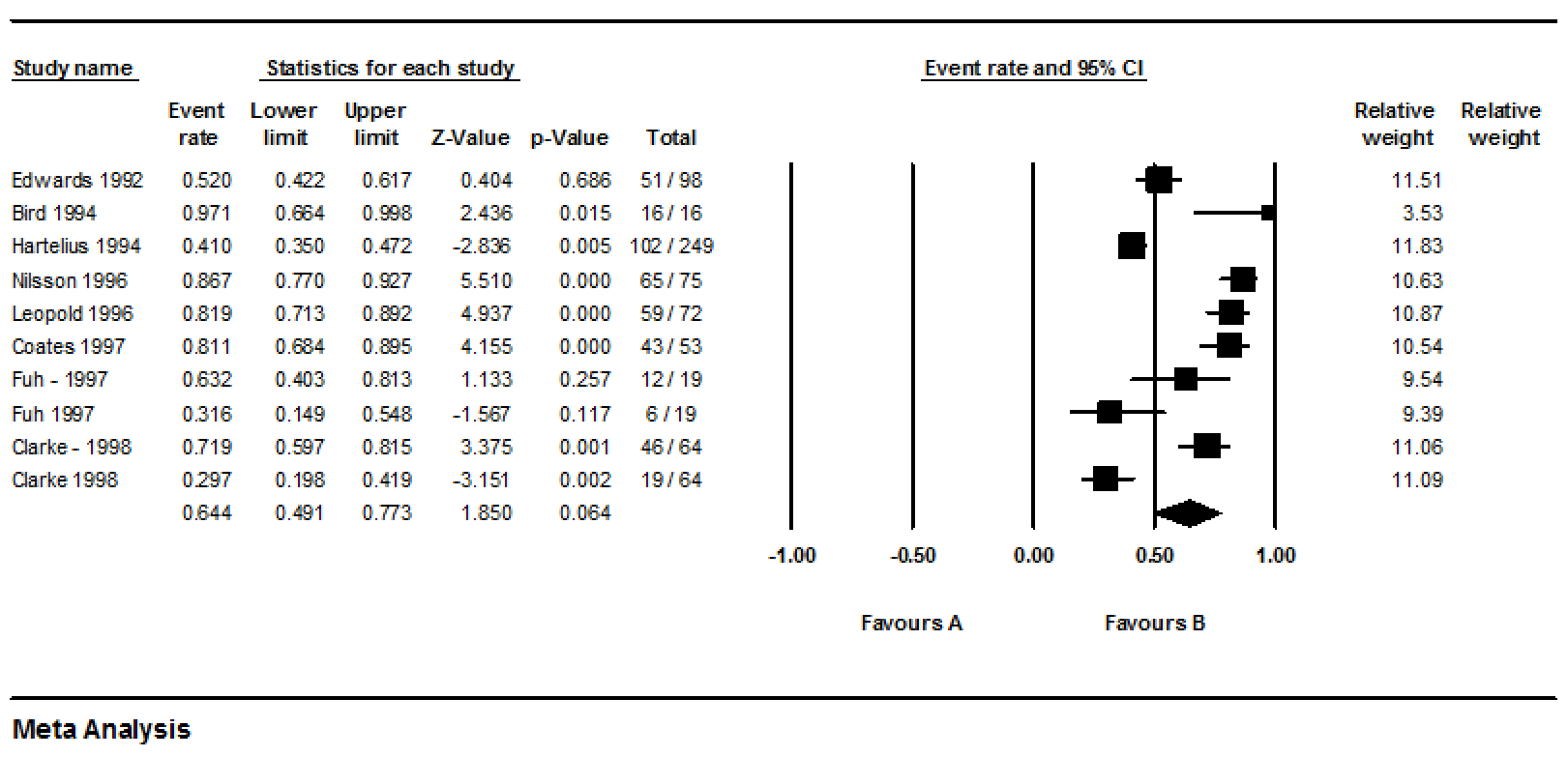
In general, according to table 2 and figure 5, the prevalence trend of dysphagia in PD decreases over time. The results of this meta-analysis show that according to 10 records (16-18, 23,24,34,35,38), the prevalence of dysphagia in Parkinson’s disease in the last 10 years of the 20th century (1991 to 2000) was 64.4% (95%CI: 49.1-77.3) (Figure 6).
According to 14 records, the prevalence of dysphagia in PD in the first 10 years of the 21st century (2001-2010) 50.1% (95%CI: 36.6-63.7) (Figure 7) (6,13,21,27,28,33,42-45,47), and in the last 10 years (2011 to 2021) was 44.7% (95%CI: 33.4-56.6) (Figure 8) according to 25 records (1, 4,9,10,15,19,20,22,25,26,29-32,36,37,39-41,46,48).
A considerable heterogeneity within all the included studies was significant (Q=2131.59, df=48, p<0.0001, I2=97.74%), supporting the use of random-effects meta-analysis. Also, the heterogeneity within the subjective group (Q=1400.22, df=29, p<0.0001, I2= 97.92%) and within the objective group (Q=252.18, df=18, p<0.0001, I2=92.86%) was significant.
Discussion
The findings of this meta-analysis provide an available overview of the prevalence of dysphagia in patients with Parkinson’s disease over three decades from 1990 to 2021. In this study, the prevalence reported in the studies was classified and reported into three categories: objective, subjective, and overall (total objective and subjective). Another finding of this study was the declining trend in prevalence of dysphagia in PD over the last three decades. The duration of PD and the evaluation method of dysphagia in these patients were two important and influential factors on the prevalence rate of dysphagia in PD. In this study, there was a significant relationship between the prevalence of dysphagia with the duration of PD and the age of patients. As the severity of PD is directly related to the age of patients and duration of PD (49), it could be expected that the increasing severity of the disease led to the increasing prevalence of dysphagia. The number of countries used to estimate the prevalence of dysphagia in PD patients was higher in this study (16 countries) than those of the previous similar study (6 countries) (50).
In general, half of the patients (50.4%) showed some types of dysphagia and the objective evaluation methods revealed higher prevalence than the subjective evaluation methods (68.8 vs. 39.5%). This finding can be explained by the various sample size of different studies. For example, the sample size in the study of Cereda et al was 6462 patients with PD (10), while Miller et al studied 137 patients with PD using the objective evaluation method (27). These findings may imply that the prevalence small target population or sampling technique has an important effect on the findings of prevalence studies (51). Another reason that can be considered for this difference is the greater accuracy of the objective evaluations tools such as Videofluoroscopic swallowing study (VFSS), FEES, and Electromyography (EMG) techniques that show accurate and complete results compared to subjective evaluation tools including interviews and self-reports (52). Also, it is possible that the patients with dysphagia are unaware of their problem (53). The Unified Parkinson’s Disease Rating Scale (UPDRS) as a subjective evaluation tool and VFSS as a gold standard for diagnosing swallowing disorders are commonly used in Parkinson’s patients (21). Out of a total of 39 studies, 10 of them used UPDRS, and 10 studies used VFSS. The high prevalence of dysphagia in an objective rather than subjective assessments (68.8 vs. 39.5%) is consistent with the findings of a previous similar study (82 vs. 23%) (50). In general, the difference between the prevalence of dysphagia in objective and subjective tools indicates that either patient with PD is unaware of their swallowing problems or does not always report their swallowing problems unless asked.
According to our study, the mean of the duration of PD was 6.8 (SD=0.7) years, and the prevalence of dysphagia increases with the increasing duration of the disease, which is in line with many studies (34,35,38). This finding may imply that dysphagia is not a clear symptom in the early stage of PD (33,34).
The next important point in this systematic review was to examine the trend in the prevalence of dysphagia in PD over a 30-year timeline. According to table 2 and figure 8, it was found that the prevalence of dysphasia in subjective studies has been almost constant for three decades and the decrease in the reported prevalence has been related to objective studies. One reason for this could be that the diagnosis of Parkinson’s disease is faster than in the past, which makes the medication and the rehabilitation process start faster and the disease progresses slower (54). The next reason is recently the awareness regarding PD and rehabilitation process of patients with PD is increased so that speech and language pathologists improve the swallowing function of patients by performing special exercises (55). Given that the prevalence of Parkinson’s disease is 22% and accounts for about 3.8% of neurological diseases and has a mortality rate of 19% (56,57), estimating the pooled prevalence of dysphagia in the PD and understanding its trend will help governments, the Parkinson’s treatment team, and the patients themselves to plan for regular evaluations and checkups, that consequently, results in timely treatment of dysphagia. The treatment of dysphagia at the early stages of PD is easier and faster and will avoid the negative consequences of dysphagia in these patients (7,8).
The normal swallowing process has four phases (preoral, oral, pharyngeal, and esophageal) (7). In contrast with the previous study, which considered only the oral and pharyngeal phases (50), in this study, we considered the dysphagia in four phases of swallowing since the impairment of them has been proven in PD patients (6-8). On the other hand, it is proved that oral or esophageal rehabilitation has been very effective for PD patients (58,59). Therefore, it is necessary to identify dysphagia in these phases and it is better to consider these two phases in estimating the pooled prevalence of dysphagia in PD.
The limitations of our study are as follows:
1) Lack of a fixed method for diagnosing the subjective and objective evaluation tools, 2) Lack of control groups in most of the included studies, 3) Lack of follow-up period in the published studies, 4) The self-reported questionnaires as a subjective evaluation tool were used in some studies, however, some PD patients were unaware of their dysphagia and did not report it unless they were asked (5,53). Lack of evaluation of the severity and stages of PD, but the duration of the PD was considered as an alternative factor.
Conclusion
In general, one in two community-dwelling PD patients is likely to have a swallowing disorder, indicating a high prevalence of dysphagia in Parkinson’s. Seven out of ten Parkinson’s patients had dysphagia through objective instruments, while four out of ten of them had dysphagia through subjective instruments. This difference suggests that subjective diagnosis tools of dysphagia may miss some PD patients with dysphagia, especially those who did not report any of swallowing complaints. Therefore, for definitive diagnosis and early treatment of dysphagia in these patients, it is recommended that after the diagnosis of Parkinson’s disease, all patients be regularly evaluated objectively for swallowing disorders.
Acknowledgement
We would like to thank Iran University of Medical Sciences for supporting the current study (ethics code: IR.IUMS.REC 1395.9211360203).
Conflict of Interest
The authors declared no conflict of interest.
References